Project Grant 2017
Deciphering the physicochemical codes for cellular function
Principal investigator:
Mikael Oliveberg, Professor of Biochemistry
Institution:
Stockholm University
Grant in SEK:
SEK 20 million over five years
Hanging on the wall of Oliveberg’s office is a photo of a ball-shaped coral. He explains that in real life it is six meters in diameter and is a whole world in itself. The biological worlds that he and his research team are exploring in their lab at Stockholm University are much smaller, but just as fascinating. They are studying cells, and in particular the physical and chemical mechanisms involved in protein function and interaction inside cells.
“If you imagine proteins as the engines of the cell, we’re the mechanics messing about with them, modifying them and tuning them, to understand how they fit together.”
With funding from the Knut and Alice Wallenberg Foundation, the project team is building on many years’ research into protein folding and aggregation, linked to neurodegenerative diseases such as ALS (also known as motor neuron disease). In recent years they have also delved into phylogenetics – the study of the relationships between organisms.
“We’re very much basic researchers. We have developed methods to address one of the great difficulties with ALS, which is to understand what happens at molecular level in the nervous system. We can now study the molecular characteristics of proteins at atomic level inside living tissue and cells. This has helped to open more doors in our research. In the biological part of the project we are studying fantastic new patterns showing the evolutionary relationship between organisms.”
Atomic movements
A human cell contains some 30,000 sorts of protein, all performing specific functions. Billions of processes involving proteins in the cell are constantly taking place.
“Cells are full of stuff – it’s like a party where you keep bumping into people, and you’re trying to find your way around. Proteins have work to do everywhere. We’re interested in how they find their way to where they need to be, which means we have to study the physics behind the processes,” Oliveberg explains.
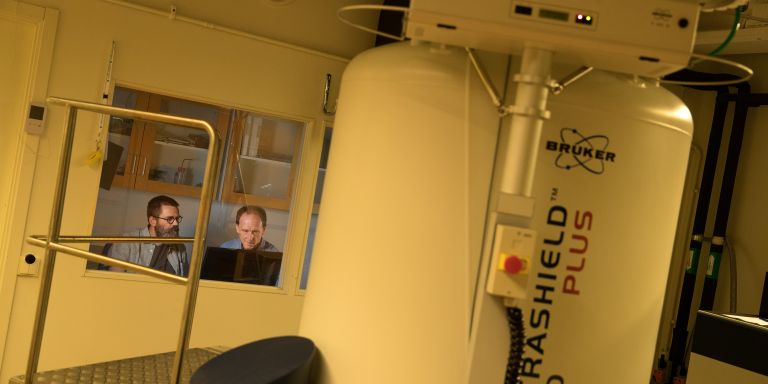
Using nuclear magnetic resonance spectroscopy (NMR), the team is able to carry out high resolution mapping of protein interaction. The proteins to be studied are dyed with various isotopes (atomic variants). The signals created via the magnetic fields in NMR enable the scientists to scan the interaction in the living cell.
“The isotope doesn’t affect how the protein works. It just creates a different magnetization so that we can see it using NMR.”
New approach to classification
The team is studying the protein’s nonfunctional surfaces. Oliveberg considers that these have been neglected in research, since they may influence the way proteins behave in the cell. Cell functions are fairly similar in biochemical terms, regardless of life form. But nonfunctional surfaces on proteins vary a great deal from one organism to another.
“Proteins must have a way of recognizing each other in the cell. That process seems to be regulated in a way that no one has seen before. They have to get up close enough to distinguish their partner, but not be so intrusive that they waste time where they don’t belong.”
Oliveberg’s team has discovered that nonfunctional surfaces regulate protein movements very precisely in cells. The surfaces have also been seen to be adapted to the environment and origin of the host cell, and its patterns can be used to classify organisms in new ways.
“When we saw this – that the proteins behave in such an orderly fashion in such a complicated system – it was fantastic. Although you might think there should a degree of order in biology, we didn’t imagine that it would be so crystal clear from a physicochemical viewpoint.”
ALS timescale
The ALS studies are continuing in parallel, using cells from “transgenic” mice, which have differing genetic variants of ALS.
In ALS, a protein in the nervous system called SOD1 mutates and forms clumps. The cells sense that something is wrong and spontaneous cell death occurs, causing the nervous system to break down. Normally, the proportion of folded and unfolded SOD1 in the cell is in equilibrium. ALS disrupts that equilibrium.
“We can predict how long the mice will live very accurately. The more unfolded protein, the more aggregation, and the more quickly the disease develops. Aggregation is almost imperceptible at first, but doubles every other week, increasing exponentially. Once symptoms appear, aggregation accelerates very rapidly, at least in mice, and then it’s too late to stop the disease.”
If this also applies in humans, SOD1 inhibitors need to be administered much earlier than is currently the case in clinical studies. Oliveberg thinks it might even be necessary before symptoms appear, perhaps as early as around the age of 40.
“To do so, it’s essential to find clinical markers for ALS – something our colleagues in Umeå are working on. The rewarding element of our ALS research is that we have gained a better understanding of the physics and chemistry involved as the disease develops in cells in mice. At molecular level everything is interconnected and follows universal rules as proteins interact.”
Text Susanne Rosén
Translation Maxwell Arding
Photo Magnus Bergström